- Visibility 171 Views
- Downloads 25 Downloads
- DOI 10.18231/j.ijceo.2021.039
-
CrossMark
- Citation
Gender, sex hormones and diabetic retinopathy: A review
- Author Details:
-
Rajendra P Maurya *
-
Ashish Gupta
-
Anup Singh
-
Virendra P Singh
-
Sanjay Bosak
-
Vibha Singh
-
Brijesh Kushwaha
-
Anil Kumar
-
Manisha
-
Eshwari Patel
Introduction
Diabetes is a chronic metabolic disorder, which has emerged as a major health care problem worldwide. In 2000, it was estimated that globally more than 171 million people had diabetes and this will grow to nearly 366 million in 2030. In India, nearly 80 million people will have adult-onset diabetes mellitus by the year 2030.[1] According to WHO, India will become one of the major hubs of diabetic population in next two decades.[2], [3] It has been reported that the rural prevalence of diabetes has increased three times in previous one and half decade (2.2% in 1989 to 6.3% in 2003).[4] Diabetes mellitus is of two types – Type 1 and Type 2. It is thought that type 1 diabetes is an autoimmune disease characterized by progressive destruction of pancreatic beta cells and insulin deficiency which is triggered by environmental factors in genetically susceptible individuals. Type 2 diabetes is a polygenic condition characterized by peripheral insulin resistance, impaired glucose metabolism by the liver and beta cell dysfunction. It is the most common type of diabetes (~90%).[5], [6]
Irrespective of type, diabetes leads to, two major vascular complications: macrovascular and microvascular. Macrovascular complications affect large vessels of the body leading to cardiovascular, cerebrovascular abnormalities and peripheral vascular disease. Microvascular complications leads to neuropathy, nephropathy and retinopathy.
Diabetic Retinopathy
The most common microvascular complication of diabetes mellitus is diabetic retinopathy (DR),[7] present in 34.6% of diabetics.[8] DR is foremost root of legitimate and functional sightlessness in the employed age group of both technologically developed and developing countries.[9], [10], [2] About 1/3rd of all diabetics, irrespective of type, develop diabetic retinopathy.[11] The incidence of diabetic retinopathy in the diabetic inhabitants increases with the duration and age of the disease. Almost every three out of four patients of diabetes are affected after 15 years of disease development.[1] DR accounts for 1 % of all visual loss and 1% of the complete blindness (year 2020). The global incidence of DR in a 10 year interval (from 1980-1982 to 1990-1992) was 74% according to Wisconsin epidemiologic study of diabetic retinopathy (WESDR).[12] The comparative risk for developing diabetic retinopathy is more in type 1 diabetes compared to type 2 diabetes.[13], [14] The prevalence of DR in type-2 DM was found to be 35-39% in the United Kingdom Prospective Diabetes Study.[15] The occurrence of diabetic retinopathy in India is increasing as the number of diabetics are increasing.[16]
Diabetic retinopathy is classified into early stage, non-proliferative diabetic retinopathy (NPDR), proliferative diabetic retinopathy (PDR) and advanced stage. . Microaneurysms, retinal haemorrhages (flame shaped, dot and blot), exudates, cotton wool spots, venous beading and looping and IRMA (intraretinal microvascular abnormalities) are associated with NPDR ([Figure 1]) and NVD (neovascularization at disc) and NVE (neovascularization elsewhere in the retina) are associated with PDR ([Figure 2] ). The risk of sight loss in the PDR is due to vitreous haemorrhage, retinal detachment and neovascular glaucoma. Diabetic macular edema (DME) remains the most common reason of visual damage in diabetic affected subjects and it can arise at any stage.[17] The rate of DME upsurges according to the period and severity of diabetes.[18]


Gender Differences in Diabetes and Diabetic Retinopathy
It has been stipulated that type 1 diabetes is more common in males than females after puberty post puberty.[19] In extension, male sex appears to have a high risk for advanced diabetic retinopathy than females.[20] However, not many of the researchers have found any gender difference for mild retinopathy or clinically significant macular edema.[21], [22] In spite of the fact that several studies of type 2 diabetes appeared to have no sex inclination in predominance or progression of diabetic retinopathy [23], [24], [25], [26], [27] but Wisconsin Epidemiological study of Diabetic Retinopathy (WESDR) appeared to show that male sex was an independent risk factor for the progression of diabetic retinopathy.[12] It was shown in the Los Angeles Latino Eye Study that in type 2 diabetes, men were more prone to develop diabetic retinopathy.[26] In India, a study also showed diabetic retinopathy has an increased risk in men who develop diabetes over forty years age.[28] There was a higher prevalence of PDR in males than females, in those patients who were having diabetes for more than 20 years duration (29% versus 9% respectively).[29] It is also seen that diabetic retinopathy status in patients of type 2 diabetes have better outcome in women than their men equivalent.[30]
In a prospective study it is reported that an additional risk factor in the development of proliferative diabetic retinopathy is commencement of diabetes before puberty.[31] Post-pubertal duration of IDDM is proposed to be more accurate determinant than total duration within the development of microvascular complications and diabetes related mortality.[32]
In spite of strict glycemic control, diabetic retinopathy presents a major issue during childbearing years. There's an increasing speed of diabetic retinopathy during pregnancy (Klein et al.1990; The Diabetic Control and Complications Trial Bunch 1993)[33], [34], it can be due to the disabled capacity of the retina to autoregulate its blood flow. [35], [36], [37], [38] The major endocrine change associated with pregnancy is extreme elevation of estrogen and progesterone.[39] The significant difference has been observed in retinal function between men and ladies below 50 years. Multifocal electroretinography studies have shown better retinal function in women of reproductive age than men’s and postmenopausal women’s.[40]
Neuroretinal Function Differences Between Males and Females
Type 1 and Type 2 diabetes, both are known to affect neural and vascular function and diabetic retinopathy followed by neuropathy. Neurodegenerative alterations in the retina precede the onset and progression of diabetic retinopathy [41], [42] and mfERG (multifocal electroretinogram) are being used to predict the situation of future retinopathy up to 3 years before clinically visible changes in vasculature occur.[43], [44], [45], [46] Electroretinography measures two parameters: Amplitude and Implicit time. mfERG speaks to the simulated activity of a large portion of the retina (103 areas) and makes a difference in spatial localization of neuroretinal dysfunction.[44]
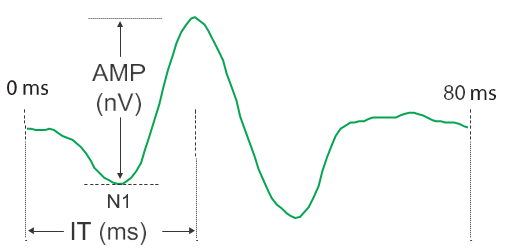
Whereas comparing nondiabetic healthy men and women beneath 50 years of age and above 50 years of age utilizing photopic mfERG, neuroretinal function of nondiabetic men and women under 50 year of age differed considerably. Under 50 years of age group, males have significantly longer implicit time than females, such difference was not shown in above 50 years of age group and there was no critical difference in amplitude response between males and females and between under and over 50 years of age subjects.[41] Utilizing ordinary flash electroretinogram (under scotopic conditions), men have smaller amplitudes than women.[47], [48], [49]
Utilizing mfERG in diabetics, it was noticed that implicit times were delayed in both the type.[50] The onset of retinopathy in both the types was not related with sex.[47] with diabetic retinopathy, it was seen that male sex was an independent risk factor for the development of macular edema.[51] Unlike the significant neuroretinal difference between nondiabetic males and females under 50 years of age, in subjects under 50 years of age with type 2 diabetes and no retinopathy, verifiable times were comparative in both the genders. In type 2 diabetes without retinopathy, the local neuroretinal function is more unusual in males compared with females,[52] recommending a sex-related protective mechanism. In animal models of ß-cell dysfunction, it has been said that estradiol ensures the pancreatic islet cells in male and ovariectomized females[53] by causing ß-cell hypertrophy, increasing insulin secretion and concentration within the pancreas.[54]
Sex Hormones
Sex hormone receptors
A few animal and human studies revealed that the eye could be a potential target for sex steroid hormones. It has been illustrated that in a large number of sex steroid hormone receptors (SSHRs) mRNA is present in ocular structures including lacrimal and meibomian glands, conjunctiva, cornea, uveal tissue and retina.[55], [56] Dihydrotestosterone (DHT) is converted from testosterone by enzyme 5 α reductase within the target cells and DHT has higher affinity for Androgen receptor (AR).[57] Estrogen has two receptors ERα and ERß, each subtype has numerous variations.[58] Progesterone receptor moreover have two subtypes – progesterone receptor A (PRA) and progesterone receptor B (PRB). AR, ERα and ERß and PRA and PRB, have been found in vascular endothelial cells, vascular smooth muscle cells (VSM), macrophages and monocytes[59], [60], [61] of all of the above mentioned visual structures. ERs although located either in the cytoplasm or on the plasma membrane are nuclear receptors.[62], [63] ER has also been seen in the vascular endothelium of organs, including the retina[57], [64], [65] ERα is localized within the layers of outer and inner nuclear layers, the outer plexiform layer, the nuclei of the ganglion cell layer and the RPE. [65] ERß, in both males and females, is localized in the RPE and neovascular tissue evolving from the choroid and is dependent on estrogen concentration.[56], [66], [67]
The distribution of sex hormone receptors in visual structures is variable and depends on sex and age of the person, which may clarify the sex differences in the epidemiology of various ocular disorders.[56] Ogueta et al. illustrated of ERα enzyme by PCR method, Western blot and immunohistochemical analysis within the retina and RPE of young ladies, but not in postmenopausal ladies or men.[64] ERα is said to have marked protective effects to vascular injury and ERß, especially in women, is believed to be the dominant form in VSM.[68] PRB seems to play a task in gene transcription and cell proliferation of VSM.[69] Gonadal hormones have gender-specific effects on lipid metabolism, obesity (central weight gain, i.e android) and glucose metabolism, explaining the differences in cardiovascular disorder (CVD) risk between men and ladies.[70], [71], [72], [73] These risk factors, along with blood pressure are also risk factors for diabetic retinopathy, which is another link between gonadal hormones and the development and progression of retinopathy.[74]
AR concentration is comparatively lower in females than in males and it is regulated by a mixture of estradiol and testosterone.[59] mRNAs of AR are found in adult rat retina and ufa, rabbit retina and choroid and human RPE.[55], [75] mRNA of 5α-reductase has also been found within the RPE,[75] it's the enzyme which converts testosterone to the more active form DHT. AR protein is found in all layers of rat retina except the ganglion and outer nuclear layers.[76]
Synthesis of steroid hormone in the retina
It has been demonstrated that the mammalian retina has the capability to synthesize neurosteroids i.e., pregnenolone, progesterone, dehydroepiandrosterone, desoxy-corticosterone, 17-hydroxyprogesterone and 17-hydroxypregenelone etc. from cholesterol with the assistance of steroidogenic proteins.[77] The common steroidogenic proteins (mRNA and protein) found within the retina are cytochrome CYP11A1 (CYP450sec) which changes over cholesterol in pregnenolone, 3-ß-hydroxysteroid dehydrogenase that changes over pregnenolone in progesterone, and cytochrome CYP19A1 (P450 aromatase) that changes over testosterone in 17-ß-estradiol.[77], [78], [79], [80] CYP450scc and aromatase enzymatic activities are the principle limiting steps for synthesis of estrogen in the retina.[81] The steroidogenic enzymes are highly present in retinal neurons, photoreceptors and glial cells.[82] The principle site of retinal steroids’ synthesis is internal nuclear layer, where the enzymes’ concentration is highest.[81]
Ocular Effects of Sex Hormones
Effect on ocular blood flow
In patients with DR the abnormal autoregulatory behavior is known to appear early. Gender-specific dissimilarities in ocular blood flow and its regulation is very little known. The increased ocular blood flow in retinal capillaries of female patients is basically due to the low rate of atherosclerosis in females. It has been demonstrated that sex hormones have particular impacts on ocular blood flow and intraocular pressure. The positive effects on ocular blood flow is by estrogen while testosterone had the opposite effect. Tucker et al. reported lower blood velocities and higher resistive indices in postmenopausal women than premenopausal (Toker et al.).[83] The vasodilatory effect of estrogen is mediated via release of nitric oxide (NO). Higher estrogen levels may lead to decreased plasma levels of a potent vasoconstrictor which is Endothelin-1 (ET-1), however, high testosterone levels associated with high plasma ET-1 level.[84]
Effects of sex hormones on blood vessels
Vascular tone
It has been demonstrated by a several large randomized studies (DCCT/EDIC Consider Inquire about Bunch 2005; The Diabetes Control and Complications Trial Gather 1993; UK Planned Diabetes Ponder Gather 1998[85], [33], [86] that lower blood pressure results in less diabetic retinopathy. It is well set up that adolescent and premenopausal women have lower blood pressure than age matched males and blood pressure rises after menopause in ladies.[87] Animal studies have shown that arterial tone and blood pressure control have involvement of ERß controlled genes.[73]
Vascular tone is controlled by gonadal hormones. Nitric Oxide (NO) may be a vasodilator and it is produced by vascular endothelial cells by endothelial nitric oxide synthase (eNOS), it at that point diffuses from endothelial cells to vascular smooth muscle where it trigger events leading to vascular dilatation.[88] Estrogen in both genomic and non-genomic pathways increases NO generation by generation of eNOS. Arterial endothelial NO release is higher in women than men.[59] Progesterone by its non-genomic pathways can lead to vascular relaxation in some tissues whereas no impact on other tissues, so its impact on vascular activity isn't clear. [59] Androgen levels are contrarily related to blood pressure,[89] it has been connected to pro-hypertensive effects. In male hypertensive rats, testosterone has been found to extend the activity of tyrosine hydroxylase, a rate limiting enzyme in norepinephrine synthesis and also increased expression of thromboxane A2 (TXA2) which ends up in increased coronary constriction.[90] These impacts appear to be produced through AR since straightened (AR blocker) disposes of the blood pressure difference between male and female mice.[89]
Lipids
Hypercholesterolemia has more chance of developing high-risk PDR and loss of vision due to diabetic macular edema (DME).[91], [92] LDL glycation or oxidation can have toxic effect on pericytes.[93] The action of hormone replacement therapy (HRT) and endogenous sex steroid hormones affect lipid levels by their action on liver lipoprotein metabolism.
Hypoandrogen metabolic disorder (HAM) is characterized by low endogenous testosterone levels in men that leads to altered lipid profile comprising of diminished HDL, expanded LDL, TG and add up to cholesterol levels and is often accompanied by metabolic disorder of central obesity, insulin resistance, hyperinsulinemia and type 2 diabetes.[89], [94] Men with higher levels of testosterone have much better lipid profile. [89] Surprisingly, supplementation of testosterone decreases HDL and influence LDL.[95] Testosterone increases expression of ß-adrenergic receptor, protein kinase A and lipase, by acting through its adipocyte androgen receptors.[96] Androgen and its blocker have mixed results in treating or preventing retinopathy.
After menopause, it is seen that HDL level decrease and LDL and TG levels increase. Lipid levels are positively affected by HRT which is seen as lowering of total cholesterol, LDL and increase of HDL, but negatively by raising triglycerides. [95] By utilizing transdermal HRT treatment, it moreover brings down TG levels as compared to oral treatment.[97] By utilizing exogenous estrogens, lipid profile gets to be better, but by using estrogen with progestin, there are increased chances of CVD (WHI and HERS). The neutral effect of progesterone is seen on lipids or also it can affect estrogen’s positive effects.[98], [99], [100]
Endothelium
The sequelae developed in diabetic retinopathy as a result of damage to capillary endothelial cells, pericytes and their surrounding basement membrane in the retina are breakdown of blood retinal barrier, basement membrane thickening and capillary obliteration.[75]
Macrophages play important roles in tissue repair but as chronic mediators of inflammation they may contribute to diabetic retinopathy.[101] In the blood vessels of diabetic patients, there is increased expression of ICAM-1 and in type 1 diabetics, soluble VCAM-1 has been detected in the serum[102], [103] Leukostasis, capillary occlusion, blood-retinal barrier disruption, increased retinal edema, endothelial cell injury and death can be correlated with increased adhesion of leukocytes to blood retinal barrier disruption.[102]
During inflammation the expression of ICAM-1 and VCAM-1 in the vascular endothelium is inhibited by estrogen[104], [105], [60] which VCAM-1 inhibition happens through ERß. This inhibition of ICAM-1 and VCAM-1 was blocked by the ER antagonist ICI 182,780, but tamoxifen, which could be a selective estrogen receptor modulator (SERM) had no inhibitory impacts.[105] The expression of VCAM-1 is inhibited by progesterone at supra-physiological levels but medroxyprogesterone acetate (MPA) does not. At normal physiological concentrations progesterone has no effect on ICAM-1 or VCAM-1 expression when tested with HUVECs.The estrogen inhibition of ICAM-1 and VCAM-1is increased by the progesterone in the human iliac artery when estrogen and progesterone are used concurrently at supra-physiological levels.[60]
Endothelial cells proliferation can be considered as a positive or negative factor. It can be positive as a vessel repair mechanism, or negative as within the tube vessel formation that happens in proliferative diabetic retinopathy or rheumatoid joint pain. An intact endothelium is vital in conditions such as atherosclerosis and diabetic retinopathy due to its role in giving “an antithrombotic and anticoagulant surface”.[62] In endothelial cell proliferation gender differences have been seen. Testosterone, increased the multiplication of vascular endothelial cells (VEC) in male rats, but had no impact on the proliferation of the VEC in female rats.[73] Progesterone acting through PR had an inhibitory impact on endothelial cell expansion in WT mice.[62]
Basement membrane
Angiogenesis remains a complex process including breakdown of a vessel wall, endothelial cell migration, expansion, tube formation and formation of a new basement membrane.[106] In this process extracellular matrix protein functions as scaffold. The potent extracellular cytokine which functions in these stages of neovascularization is basic fibroblast growth factor (FGF-2). FGF-2 manufactured in endothelial cell is unique as it lacks a sequence to allow its release from the endothelial cell. In basement membrane remodeling extracellular matrix proteases which are regulated post translationally play an important role. Plasminogen activators (tPA and uPA) are inhibited by the plasminogen activator inhibitor (PAI-1).When endothelial cells are tranquil, such as when they are confluent, estrogen diminishes protease movement, by expanding PAI-1 generation and diminishing TPA and uPA generation. When cells are activated, such as in angiogenesis, estrogen accelerates protease activity, by decreasing PAI-1 generation and subsequently expanding tPA and uPA generation.[107]
Pericytes
Pericytes and VSM both are derived from the same pleuripotent cells.[102] They play a part in supporting the microcirculation against hydrostatic pressure and through physical and chemical signaling along with vessel endothelial cells they also function to support vessel stability.[108] The first pathological microscopic damage associated with hyperglycemia is pericyte loss.[109], [110] Utilizing bovine retinal capillary pericytes, it was found that DHEA, a precursor to both testosterone and estrogen, decreases pericyte loss resulting from high glucose. It was also reported that this impact was not due to either AR or ER as nanomolar concentrations of DHT and E2 had no effect on pericyte loss resulting from high glucose.[109], [110]. The antioxidant properties are responsible for the protective cellular effect of DHEA.[111], [112]
Angiogenesis
The most important inhibitor of angiogenesis in mammalian eyes is pigment epithelium-derived factor (PEDF), is a glycoprotein (50-kDa) secreted by the RPE,[112], [113] suggesting that in antigenic eye diseases such as proliferative diabetic retinopathy decreased level of PEDF plays an important role.[114], [115] In a study on rats, it is found that VEFG levels are inversely related to retinal oxygen concentrations and PEDF levels in a balanced controlling angiogenesis.[116] A study on human ovarian and surface epithelial cells found that E2 reduces PEDF levels and this reduction can be balanced by the presentation of the ER antagonist, ICI 182,780, recommending that ER is a vital upstream controller of PEDF.[117] Selective estrogen receptor modulators (SERMs), counting tamoxifen and raloxifene, can act as agonists or antagonist of estrogen depending upon the receptor (ER) or coactivators present in a specific cell. They can be outlined in such a way that they only suppress that ER which is found in retinal microvascular endothelial cells, so as to reduce leukostasis by inhibiting ICAM-1 or VCAM-1[116] and their activities can be either genomic or non-genomic. It is seen that in inhibition of estrogen mediated angiogenesis raloxifene and tamoxifen have similar potency.[118]
Hormone Replacement Therapy, Menopause and Diabetes
The risk of developing diabetes or diabetic retinopathy is altered by many intrinsic and extrinsic differences between males and females. HRT in type 2 diabetes is one of the most important factors. In postmenopausal women, HRT in the frame of conjugated equine estrogen or conjugated equine estrogen additionally medroxyprogesterone acetic acid derivation decreased the risk of diabetes and on discontinuing HRT, chances of reduction of diabetes vanishes. [119] There was a 35% risk reduction seen in diabetes in a study where women with coronary artery disease were taking conjugated estrogen and medroxyprogesterone acetate.[120]
Menopause is another thought. Universally, under 60 years of age, diabetes predominance is marginally higher in males, but in more older ages, it is marginally higher in females.[1] Menopause earlier in life, naturally or surgically (hysterectomy with respective oophorectomy) was related with a essentially higher risk for develoment of diabetes,[121] may be the due to sudden withdrawal of estrogen during menopause suggesting its protective nature in females. One study reported higher rate of diabetic blindness in males beneath 45 years of age than females under 45 years of age and the rate was comparable in males and females above 45 years of age.[122]
Role of Sex Hormone in Diabetic Retinopathy
It is accepted that in diabetic retinopathy, hyperglycemia induced retinal damage occurs through oxidative stress initiated through various mechanisms[123], [73], [124]. The glucose toxicity and associated vascular damage is believed to occur through four ways: 1) activation of protein kinase C (PKC), 2) enactment of the protein aldose reductase, 3) accumulation of advanced glycation end product (AGE) and 4) activation of the hexosamine pathway. “Unifying hypothesis” recommends that formation of reactive oxygen species (ROS) is the result of each of these mechanism driving to glucose toxicity.[74], [123]
As oxidative stress is responsible for the development and progression of diabetic retinopathy, estrogen comes into play because it has anti-oxidant properties. ARPE-19 cells express both the ERα and ERß. 17-ß estradiol (17ß-E2) decreases ROS levels and mitochondrial depolarization in ARPE-19 cells and this cytoprotection was inhibited by ER antagonists ICI (ERα and ERß) and THC (ERß) but not by tamoxifen (ERα). This affirms that Estradiol (17ß-E2) protects ARPE-19 cells from oxidative damage and apoptosis through ERß dependent mechanisms i.e., by maintenance of mitochondrial function, reducing ROS levels and expression of cellular antioxidant genes.[66], [124]
It has been proven that gonadal hormones do play a part in development and progression of diabetic retinopathy but treatment of diabetic retinopathy utilizing gonadal hormones, or their blockers, cannot be bound together for all the patients. Suitable gonadal hormonal incitement or treatment can give a novel helpful alternative for diabetic retinopathy.
Source of Funding
None.
Conflict of Interest
None.
References
- S Wild, G Roglic, A Green, R Sicree, H King. Global Prevalence of Diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004. [Google Scholar]
- H King, R E Aubert, W H Herman. Global Burden of Diabetes, 1995-2025: Prevalence, numerical estimates, and projections. Diabetes Care 1998. [Google Scholar]
- R P Maurya. Diabetic retinopathy: My brief synopsis. Ind J Clin Exp Ophth 2015. [Google Scholar]
- A Ramachandran, M V Jali, V Mohan, C Snehalatha, M Viswanathan. High prevalence of diabetes in an urban population in south India.. BMJ 1988. [Google Scholar]
- . World Health Organization. 10 Facts about diabetes. 2013. [Google Scholar]
- D M Maahs, N A West, J M Lawrence, E J M Davis. Epidemiology of Type 1 Diabetes. Endocrinol Metab Clin North Am 2010. [Google Scholar]
- S Sivaprasad, B Gupta, R C Nwaobi, J Evans. Prevalence of Diabetic Retinopathy in Various Ethnic Groups: A Worldwide Perspective. Surv Ophthalmol 2012. [Google Scholar]
- J W Y Yau, S L Rogers, R Kawasaki, E L Lamoureux, J W Kowalski, T Bek, . Global Prevalence and Major Risk Factors of Diabetic Retinopathy. Diabetes Care 2012. [Google Scholar]
- N G Congdon. Important Causes of Visual Impairment in the World Today. JAMA 2003. [Google Scholar]
- D S Fong, L P Aiello, F L Ferris, R Klein. Diabetic Retinopathy. Diabetes Care 2004. [Google Scholar]
- N Cheung, P Mitchell, T Y Wong. Diabetic retinopathy. The Lancet 2010. [Google Scholar]
- R Klein, K E Lee, R E Gangnon, B E K Klein. The 25-Year Incidence of Visual Impairment in Type 1 Diabetes Mellitus. Ophthalmology 2010. [Google Scholar]
- M S Roy, R Klein, B J O’colmain, B E Klein, S E Moss, J H Kempen. The prevalence of diabetic retinopathy among adult type 1 diabetic persons in the United States. Arch Ophthalmol 2004. [Google Scholar]
- H P Hammes, W Kerner, S Hofer, O Kordonouri, K Raile, R W Holl. Diabetic retinopathy in type 1 diabetes—a contemporary analysis of 8,784 patients. Diabetologia 2011. [Google Scholar]
- E M Kohner, S J Aldington, I M Stratton, S E Manley, R R Holman, D R Matthews. United Kingdom Prospective Diabetes Study, 30: diabetic retinopathy at dignosis of non-insulin-dependent diabetes mellitus and associated risk factors. Arch Ophthalmol 1998. [Google Scholar]
- L Dandona, R Dandona, R K John. Estimation of blindness in India from 2000 through 2020: implications for the blindness control policy. Natl Med J India 2001. [Google Scholar]
- P Romero, M Baget, I Mendez, J Fernández, M Salvat, I Martinez. Diabetic macular edema and its relationship to renal microangiopathy: a sample of Type I diabetes mellitus patients in a 15-year follow-up study. J Diabetes Complications 2007. [Google Scholar]
- R P Maurya. Dabetic macular edema: An overview. Indian J Clin Exp Ophthalmol 2019. [Google Scholar]
- N Lammi, O Taskinen, E Moltchanova, I L Notkola, J G Eriksson, J Tuomilehto. A high incidence of type 1 diabetes and an alarming increase in the incidence of type 2 diabetes among young adults in Finland between 1992 and 1996. Diabetologia 2007. [Google Scholar]
- H J Bodansky, A G Cudworth, R A Whitelocke, J H Dobree. Diabetic retinopathy and its relation to type of diabetes: review of a retinal clinic population.. Br JOphthalmol 1982. [Google Scholar]
- R Klein, M D Knudtson, K E Lee, R Gangnon, B E K Klein. The Wisconsin Epidemiologic Study of Diabetic Retinopathy XXIII: The Twenty-five-Year Incidence of Macular Edema in Persons with Type 1 Diabetes. Ophthalmology 2009. [Google Scholar]
- N Chaturvedi, A K Sjoelie, M Porta, S J Aldington, J H Fuller, M Songini. Markers of Insulin Resistance Are Strong Risk Factors for Retinopathy Incidence in Type 1 Diabetes: The EURODIAB Prospective Complications Study. Diabetes Care 2001. [Google Scholar]
- T S T Smith, J Szetu, R R A Bourne. The prevalence and severity of diabetic retinopathy, associated risk factors and vision loss in patients registered with type 2 diabetes in Luganville, Vanuatu. Br J Ophthalmol 2007. [Google Scholar]
- R J Tapp, P Z Zimmet, C A Harper, D J McCarty, P Chitson, A M Tonkin. Six year incidence and progression of diabetic retinopathy: Results from the Mauritius diabetes complication study. Diabetes Res Clin Pract 2006. [Google Scholar]
- F H Wang, Y B Liang, F Zhang, J J Wang, W B Wei, Q S Tao. Prevalence of diabetic retinopathy in rural China: the Handan Eye Study. Ophthalmology 2009. [Google Scholar]
- T Y Wong, N Cheung, W T Tay, J J Wang, T Aung, S M Saw. Prevalence and risk factors for diabetic retinopathy: the Singapore Malay Eye Study. Ophthalmology 2008. [Google Scholar]
- T Y Wong, R Klein, F M A Islam, M F Cotch, A R Folsom, B E K Klein. Diabetic Retinopathy in a Multi-ethnic Cohort in the United States. Am J Ophthalmol 2006. [Google Scholar]
- R Raman, K Vaitheeswaran, K Vinita, T Sharma. Is Prevalence of Retinopathy Related to the Age of Onset of Diabetes? Sankara Nethralaya Diabetic Retinopathy Epidemiology and Molecular Genetic Report No. 5. Ophthalmic Res 2011. [Google Scholar]
- D K Nagi, D J Pettitt, P H Bennett, R Klein, W C Knowler. Diabetic retinopathy assessed by fundus photography in Pima Indians with impaired glucose tolerance and NIDDM. Diabet Med 1997. [Google Scholar]
- R Klein, B E Klein, S E Moss, K J Cruickshanks. The Wisconsin Epidemiologic Study of diabetic retinopathy. XIV. Ten-year incidence and progression of diabetic retinopathy. Arch Ophthalmol 1994. [Google Scholar]
- M Porta, A K Sjoelie, N Chaturvedi, L Stevens, R Rottiers, M Veglio. Risk factors for progression to proliferative diabetic retinopathy in the EURODIAB Prospective Complications Study. Diabetologia 2001. [Google Scholar]
- J N Kostraba, J S Dorman, T J Orchard, D J Becker, Y Ohki, D Ellis. Contribution of Diabetes Duration Before Puberty to Development of Microvascular Complications in IDDM Subjects. Diabetes Care 1989. [Google Scholar]
- . The Diabetes Control and Complications Trial. Diabetes Control Complications Trial 1993. [Google Scholar]
- B E K Klein, S E Moss, R Klein. Effect of Pregnancy on Progression of Diabetic Retinopathy. Diabetes Care 1990. [Google Scholar]
- J T Ernest, T K Goldstick, R L Engerman. Hyperglycemia impairs retinal oxygen autoregulation in normal and diabetic dogs. Invest Ophthalmol Vis Sci 1983. [Google Scholar]
- J E Grunwald, C E Riva, A J Brucker, S H Sinclair, B L Petrig. Altered Retinal Vascular Response to 100% Oxygen Breathing in Diabetes Mellitus. Ophthalmology 1984. [Google Scholar]
- J E Grunwald, C E Riva, B L Petrig, A J Brucker, S S Schwartz, S N Braunstein. Strict control of glycaemia: effects on blood flow in the large retinal vessels and in the macular microcirculation.. BrJOphthalmol 1995. [Google Scholar]
- S M Rassam, V Patel, E M Kohner. The effect of experimental hypertension on retinal vascular autoregulation in humans: a mechanism for the progression of diabetic retinopathy. Exp Physiol 1995. [Google Scholar]
- L J Peterson, C M Peterson. Diabetic retinopathy. Clin Obstet Gynecol 1991. [Google Scholar]
- G Y Ozawa, M A Bearse, W W Harrison, K W BCastain, M E Schneck, S Barez. Differences in Neuroretinal Function between Adult Males and Females. Optom Vis Sci 2014. [Google Scholar]
- AYoshida, M Kojima, H Ogasawara, Satoshi Ishiko. Oscillatory Potentials and Permeability of the Blood-retinal Barrier in Noninsulin-dependent Diabetic Patients without Retinopathy. Ophthalmology 1991. [Google Scholar]
- G H Bresnick, M Palta. Predicting Progression to Severe Proliferative Diabetic Retinopathy. Arch Ophthalmol 1987. [Google Scholar]
- M A Bearse, Anthony J. Adams, Y Han, M E Schneck, J Ng, K B Castain. A multifocal electroretinogram model predicting the development of diabetic retinopathy. Prog Retin Eye Res 2006. [Google Scholar]
- Y Han, ME Schneck, M A Bearse, S Barez, C H Jacobsen, N P Jewell. Formulation and Evaluation of a Predictive Model to Identify the Sites of Future Diabetic Retinopathy. Invest Ophthalmol Vis Sci 2004. [Google Scholar]
- J S Ng, M A Bearse, M E Schneck, S Barez, A J Adams. Local Diabetic Retinopathy Prediction by Multifocal ERG Delays over 3 Years. Invest Ophthalmol Vis Sci 2008. [Google Scholar]
- W W Harrison, M A Bearse, J S Ng, N P Jewell, S Barez, D Burger. Multifocal Electroretinograms Predict Onset of Diabetic Retinopathy in Adult Patients with Diabetes. Invest Ophthalmol Vis Sci 2011. [Google Scholar]
- D G Birch, J L Anderson. Standardized full-field electro- retinography. Normal values and their variation with age. Arch Ophthalmol 1992. [Google Scholar]
- B V Mattila. The clinical electroretinogram; II. The difference between the electroretinogram in men and in women. Acta Ophthalmol 1951. [Google Scholar]
- I Zeidler. The clinical electroretinogram. IX. The normal electroretinogram. Value of the b-potential in different age groups and its differenes in men and women. Acta Ophthalmol 1959. [Google Scholar]
- B Fortune, M E Schneck, A J Adams. Multifocal electro- retinogram delays reveal local retinal dysfunction in early diabetic retinopathy. Invest Ophthalmol Vis Sci 1999. [Google Scholar]
- W W Harrison, M A Bearse, M E Schneck, B E Wolff, N P Jewell, S Barez. Prediction, by Retinal Location, of the Onset of Diabetic Edema in Patients with Nonproliferative Diabetic Retinopathy. Invest Ophthalmol Vis Sci 2011. [Google Scholar]
- G Y Ozawa, M A Bearse, K W B Castain, W W Harrison, M E Schneck, S Barez. Neurodegenerative Differences in the Retinas of Male and Female Patients with Type 2 Diabetes. Invest Ophthalmol Vis Sci 2012. [Google Scholar]
- S Liu, F M Jarvis. Estrogenic protection of beta-cell failure in metabolic diseases. Endocrinology 2010. [Google Scholar]
- N V Costrini, R K Kalkhoff. Relative effects of pregnancy, estradiol, and progesterone on plasma insulin and pancreatic islet insulin secretion. J Clin Invest 1971. [Google Scholar]
- L A Wickham, J Gao, I Toda, EM Rocha, M Ono, D A Sullivan. Identification of androgen, estrogen and progesterone receptor mRNAs in the eye. Acta Ophthalmol Scand 2000. [Google Scholar]
- P D Gupta, K Johar, K Nagpal, AR Vasavada. Sex Hormone Receptors in the Human Eye. Surv Ophthalmol 2005. [Google Scholar]
- J I McGinley, W J Canovatchel. Complete androgen insensitivityPathophysiology, diagnosis, and management. Trends Endocrinol Metab 1992. [Google Scholar]
- J M Orshal, R A Khalil. Gender, sex hormones, and vascular tone. Am J Physiol Regul Integr Comp Physiol 2004. [Google Scholar]
- A C Villablanca, M Jayachandran, C Banka. Atherosclerosis and sex hormones: current concepts. Clin Sci 2010. [Google Scholar]
- J Thompson, R A Khalil. Gender differences in the regulation of vascular tone. Clin Exp Pharmacol Physiol 2003. [Google Scholar]
- F Vázquez, J C R Manzaneque, J P Lydon, D P Edwards, B W O'Malley, M L I Arispe. Progesterone Regulates Proliferation of Endothelial Cells. J Biol Chem 1999. [Google Scholar]
- D C Márquez, R J Pietras. Membrane-associated binding sites for estrogen contribute to growth regulation of human breast cancer cells. Oncogene 2001. [Google Scholar]
- T Simoncini, A H Moghadam, D P Brazil, K Ley, W W Chin, J K Liao. Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3-OH kinase. Nature 2000. [Google Scholar]
- S B Ogueta, S D Schwartz, C K Yamashita, D B Farber. Estrogen receptor in the human eye: influence of gender and age on gene expression. Invest Ophthalmol Vis Sci 1999. [Google Scholar]
- I Suzuma, M Mandai, H Takagi, K Suzuma, A Otani, H Oh. 17 Beta- estradiol increases VEGF receptor-2 and promotes DNA synthesis in retinal microvascular endothelial cells. Invest Ophthalmol Vis Sci 1999. [Google Scholar]
- A Giddabasappa, M Bauler, M Yepuru, E Chaum, J T Dalton, J Eswaraka. 17- beta estradiol protects ARPE-19 cells from oxidative stress through estrogen receptor-beta. Invest Ophthalmol Vis Sci 2010. [Google Scholar]
- M E M Castano, S J Elliot, M Potier, M Karl, L J Striker, G E Striker. Regulation of Estrogen Receptors and MMP-2 Expression by Estrogens in Human Retinal Pigment Epithelium. Invest Ophthalmol Vis Sci 2003. [Google Scholar]
- M E Mendelsohn. Genomic and nongenomic effects of estrogen in the vasculature. Am J Cardiol 2002. [Google Scholar]
- D Pieber. Interactions between progesterone receptor isoforms in myometrial cells in human labour. Mol Hum Reprod 2001. [Google Scholar]
- C Carani, K Qin, MSimoni, M F Fustini, S Serpente, J Boyd. Effect of Testosterone and Estradiol in a Man with Aromatase Deficiency. N Engl J Med 1997. [Google Scholar]
- V A Fonseca, Vivian Fonseca. . Cardiovascular Endocrinology. Contemporary Endocrinology 2009. [Google Scholar]
- J Liu, S Wu, H Wei, K Zhou, Y Ruan, WLai. Effects of Sex Hormones and Their Balance on the Proliferation of Rat Vascular Endothelial Cells. Horm Res 2002. [Google Scholar]
- C Vitale, M Fini, G Speziale, S Chierchia. Gender differences in the cardiovascular effects of sex hormones. Fundam Clin Pharmacol 2010. [Google Scholar]
- J C Vaz. Diabetic Retinopathy. Hackensack. 2011. [Google Scholar]
- E M Rocha, L A Wickham, L A Da Silveira, K L Krenzer, F S Yu, I Toda. Identification of androgen receptor protein and 5alpha-reductase mRNA in human ocular tissues. Br J Ophthalmol 2000. [Google Scholar]
- A Prabhu, Q Xu, M B Manigrasso, M Biswas, E Flynn, R Iliescu. Expression of aromatase, androgen and estrogen receptors in peripheral target tissues in diabetes. Steroids 2010. [Google Scholar]
- P Guarnieri, R Guarnieri, C Cascio, P Pavasant, F Piccoli, V Papadopoulos. Neurosteoidogenesis in rat retina. J Neurochem 1994. [Google Scholar]
- C Cascio, D Russo, G Drago, G Galizzi, R Passantino, R Guarneri. 17 β-Estradiol synthesis in the adult male rat retina. Exp Eye Res 2007. [Google Scholar]
- M Ishikawa, T Yoshitomi, C F Zorumski, Y Izumi. Neurosteroids Are Endogenous Neuroprotectants in an Ex Vivo Glaucoma Model. Invest Ophthalmol Vis Sci 2014. [Google Scholar]
- S J Fliesler, L Bretillon. The ins and outs of cholesterol in the vertebrate retina. J Lipid Res 2010. [Google Scholar]
- C Cascio, I Deidda, D Russo, P Guarneri. The estrogenic retina: The potential contribution to healthy aging and age-related neurodegenerative diseases of the retina. Steroids 2015. [Google Scholar]
- S H Mellon, L D Griffin, N A Compagnone. Biosynthesis and action of neurosteroids. Brain Res Brain Res Rev 2001. [Google Scholar]
- E Toker, Ö Yenice, I Akpinar, E Aribal, H Kazokoglu. The influence of sex hormones on ocular blood flow in women. Acta Ophthalmol Scand 2003. [Google Scholar]
- N T Malan, W Smith, R V Känel, M Hamer, A E Schutte, L Malan. Low serum testosterone and increased diastolic ocular perfusion pressure: a risk for retinal microvasculature. Vasa 2015. [Google Scholar]
- D M Nathan, P A Cleary, J Y C Backlund, S M Genuth, J M Lachin, T J Orchard. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005. [Google Scholar]
- . Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998. [Google Scholar]
- R K Dubey, S Oparil, B Imthurn, E K Jackson. Sex hormones and hypertension. Cardiovasc Res 2002. [Google Scholar]
- M Kaushik, S P Sontineni, C Hunter. Cardiovascular disease and androgens: A review. Int J Cardiol 2010. [Google Scholar]
- E Y Chew, M L Klein, F L Ferris, N A Remaley, R P Murphy, K Chantry. Association of elevated serum lipid levels with retinal hard exudate in diabetic retinopathy. Early Treatment Diabetic Retinopathy Study (ETDRS) Report 22. Arch Ophthalmol 1996. [Google Scholar]
- J M Berg, J L Tymoczko, L Stryer. Biochemistry. 2007. [Google Scholar]
- B E Klein, S E Moss, R Klein, T S Surawicz. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. XIII. Relationship of serum cholesterol to retinopathy and hard exudate. Ophthalmology 1991. [Google Scholar]
- T A Chowdhury, D Hopkins, P M Dodson, G C Vafidis. The role of serum lipids in exudative diabetic maculopathy: is there a place for lipid lowering therapy?. Eye 2002. [Google Scholar]
- C M Webb, P Collins. Testosterone and coronary artery disease in men. Maturitas 2010. [Google Scholar]
- M E Mendelsohn. Molecular and Cellular Basis of Cardiovascular Gender Differences. Science 2005. [Google Scholar]
- F C W Wu, A V Eckardstein. Androgens and Coronary Artery Disease. Endocr Rev 2003. [Google Scholar]
- S Nanda, N Gupta, H C Mehta, K Sangwan. Effect of oestrogen replacement therapy on serum lipid profile. Aust N Z J Obstet Gynaecol 2003. [Google Scholar]
- J E Rossouw, G L Anderson, R L Prentice, A Z Lacroix, C Kooperberg, M L Stefanick. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA 2002. [Google Scholar]
- S Hulley, D Grady, T Bush, C Furberg, D Herrington, B Riggs. Randomized Trial of Estrogen Plus Progestin for Secondary Prevention of Coronary Heart Disease in Postmenopausal Women. Obstetrical Gyneco Surv 1999. [Google Scholar]
- S L Mumford, S Dasharathy, A Z Pollack, E F Schisterman. Variations in lipid levels according to menstrual cycle phase: clinical implications. Clin Lipidology 2011. [Google Scholar]
- E Duh. . Diabetic Retinopathy. Contemporary Diabetes 2009. [Google Scholar]
- H P Hammes, M Porta. . Experimental Approaches to Diabetic Retinopathy 2010. [Google Scholar]
- K Miyamoto, S Khosrof, S E Bursell, R Rohan, T Murata, A C Clermont. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci U S A 1999. [Google Scholar]
- M C Cid, H K Kleinman, D S Grant, H W Schnaper, A S Fauci, G S Hoffman. Estradiol enhances leukocyte binding to tumor necrosis factor (TNF)-stimulated endothelial cells via an increase in TNF-induced adhesion molecules E-selectin, intercellular adhesion molecule type 1, and vascular cell adhesion molecule type 1.. J Clin Invest 1994. [Google Scholar]
- T Simoncini, R D Caterina, A R Genazzani. Selective Estrogen Receptor Modulators: Different Actions on Vascular Cell Adhesion Molecule-1 (VCAM-1) Expression in Human Endothelial Cells. J Clin Endocrinol Metab 1999. [Google Scholar]
- J Folkman. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1995. [Google Scholar]
- G M Rubanyi, R Kauffman. . Estrogen and the Vessel Wall. The Endothelial Cell Research Series 1998. [Google Scholar]
- A P Hall. Review of the pericyte during angiogenesis and its role in cancer and diabetic retinopathy. Toxicol Pathol 2006. [Google Scholar]
- H P Hammes. Pericytes and the pathogenesis of diabetic retinopathy. Horm Metab Res 2005. [Google Scholar]
- E K Vidro, S Gee, R Unda, J X Ma, A Tsin. Glucose and TGFβ2 Modulate the Viability of Cultured Human Retinal Pericytes and Their VEGF Release. Curr Eye Res 2008. [Google Scholar]
- E Brignardello, E Beltramo, PA Molinatti, M Aragno, V Gatto, E Tamagno. Dehydroepiandrosterone protects bovine retinal capillary pericytes against glucose toxicity. J Endocrinol 1998. [Google Scholar]
- M Aragno, G Meineri, I Vercellinatto, P Bardini, S Raimondo, P G Peiretti. Cardiac impairment in rabbits fed a high-fat diet is counteracted by dehydroepiandrosterone supplementation. Life Sci 2009. [Google Scholar]
- J T Tink, G G Chader, L V Johnson. PEDF: A pigment epithelium-derived factor with potent neuronal differentiative activity. Exp Eye Res 1991. [Google Scholar]
- J T Tink, L V Johnson. Neuronal differentiation of retinoblastoma cells induced by medium conditioned by human RPE cells. Invest Ophthalmol Vis Sci 1989. [Google Scholar]
- D W Dawson, O V Volpert, P Gillis, S E Crawford, H Xu, W Benedict. Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science 1999. [Google Scholar]
- K Takenaka, S Yamagishi, Y Jinnouchi, K Nakamura, T Matsui, T Imaizumi. Pigment epithelium-derived factor (PEDF)-induced apoptosis and inhibition of vascular endothelial growth factor (VEGF) expression in MG63 human osteosarcoma cells. Life Sci 2005. [Google Scholar]
- G Gao, Y Li, D Zhang, S Gee, Craig Crosson, J X Ma. Unbalanced expression of VEGF and PEDF in ischemia-induced retinal neovascularization. FEBS Letters 2001. [Google Scholar]
- L W T Cheung, S C L Au, A N Y Cheung, HY S Ngan, J T Tink, N Auersperg. Pigment Epithelium-Derived Factor Is Estrogen Sensitive and Inhibits the Growth of Human Ovarian Cancer and Ovarian Surface Epithelial Cells. Endocrinology 2006. [Google Scholar]
- J G Grigsby, K Parvathaneni, M A Almanza, AM Botello, AA Mondragon, D M Allen. Effects of Tamoxifen Versus Raloxifene on Retinal Capillary Endothelial Cell Proliferation. J Ocul Pharmacol Ther 2011. [Google Scholar]
- J E Manson, R T Chlebowski, M L Stefanick, A K Aragaki, J E Rossouw, R L Prentice. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA 2013. [Google Scholar]
- A M Kanaya, D Herrington, E Vittinghoff, F Lin, D Grady, V Bittner. Glycemic Effects of Postmenopausal Hormone Therapy: The Heart and Estrogen/progestin Replacement Study: A Randomized, Double-Blind, Placebo-Controlled Trial. Ann Intern Med 2003. [Google Scholar]
- D Appiah, S J Winters, C A Hornung. Bilateral Oophorectomy and the Risk of Incident Diabetes in Postmenopausal Women. Diabetes Care 2014. [Google Scholar]
- K K Yuen, H A Kahn. The Association of Female Hormones with Blindness from Diabetic Retinopathy. Am J Ophthalmol 1976. [Google Scholar]
- M Brownlee. Biochemistry and molecular cell biology of diabetic complications. Nature 2001. [Google Scholar]
- X L Du, D Edelstein, L Rossetti, I G Fantus, H Goldberg, F Ziyadeh. Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc Natl Acad Sci U S A 2000. [Google Scholar]
- Introduction
- Diabetic Retinopathy
- Gender Differences in Diabetes and Diabetic Retinopathy
- Neuroretinal Function Differences Between Males and Females
- Sex Hormones
- Ocular Effects of Sex Hormones
- Hormone Replacement Therapy, Menopause and Diabetes
- Role of Sex Hormone in Diabetic Retinopathy
- Source of Funding
- Conflict of Interest
